Introduction
Narcolepsy Type 1 (Narcolepsy with Cataplexy) is a neurological sleep disorder categorized by rapid onset rapid eye movement (REM) sleep and Cataplexy which is sudden paralysis when experiencing strong or sudden emotion. Patients report excessive daytime sleepiness (EDS) and frequently need naps and stimulant medication to function in day-to-day life.
HCRT and Narcolepsy Type 1
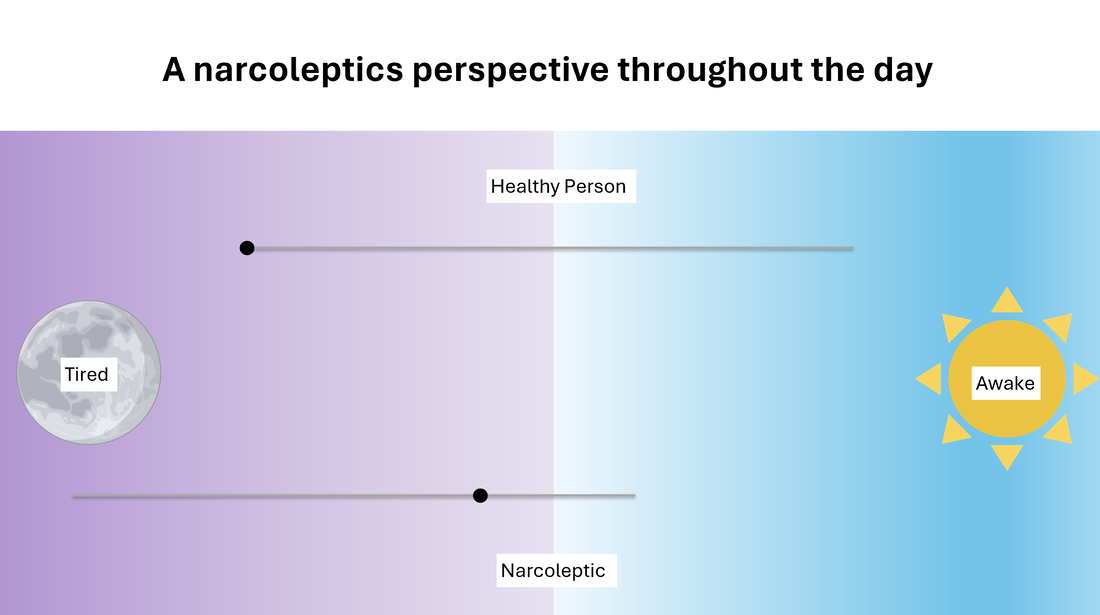
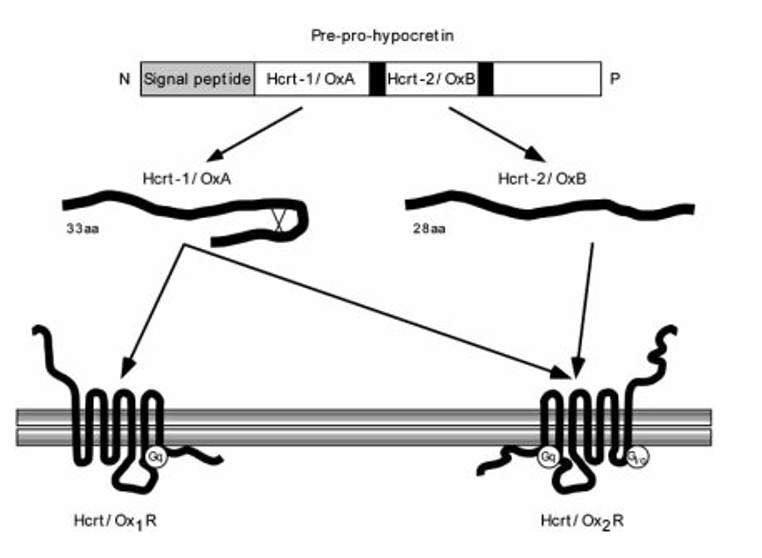
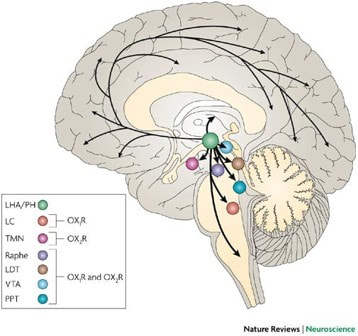
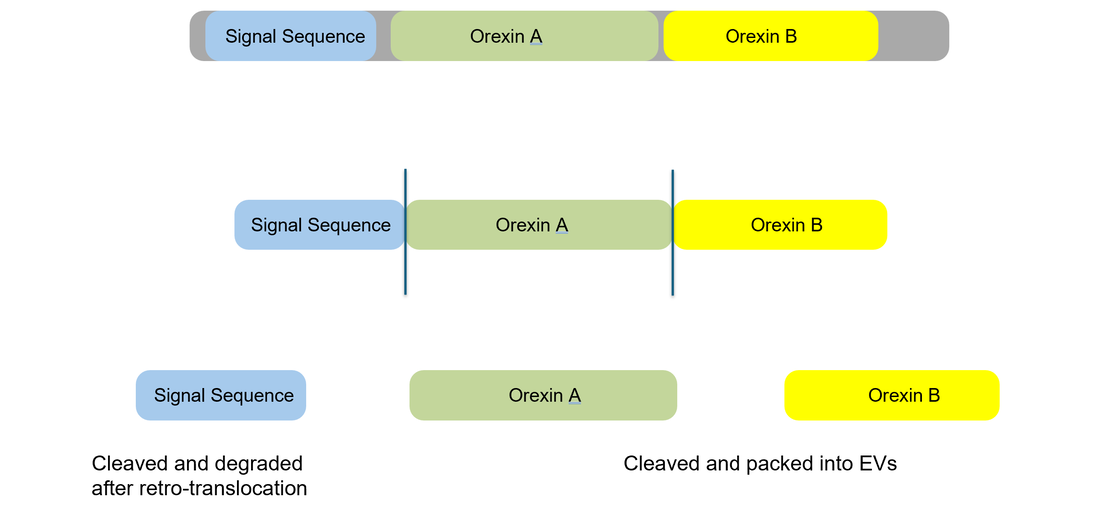
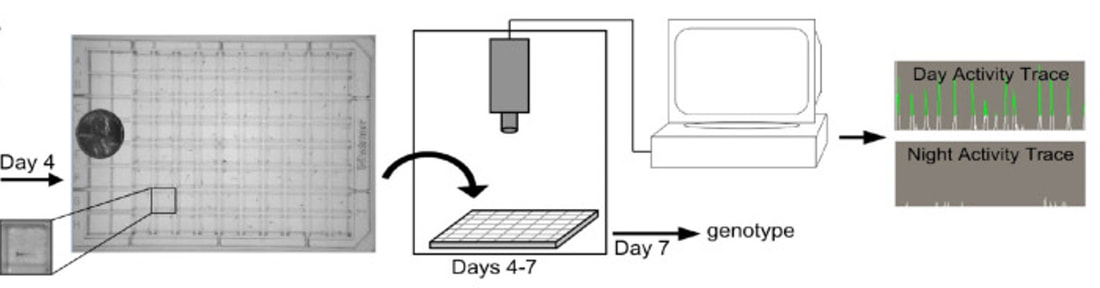
The origins of Narcolepsy were discovered in a dog model containing a mutated version of the prepo-orexin (HCRT) gene which acts as a precursor to neurotransmitter peptides orexin A and orexin B. These peptides are expressed in specific neurons that control circadian rhythm and are also thought to be the victim of an autoimmune reaction caused by infection. The absence of these peptides in the brain is thought to be the root cause of narcolepsy type 1 which changes the the feeling of "wakefulness" permanently. Narcoleptics have a hard time staying awake throughout the day and a hard time staying asleep throughout the night as shown below.
Gene Ontology
Molecular Function Cellular Component Biological Processes
|
•neuropeptide hormone activity
|
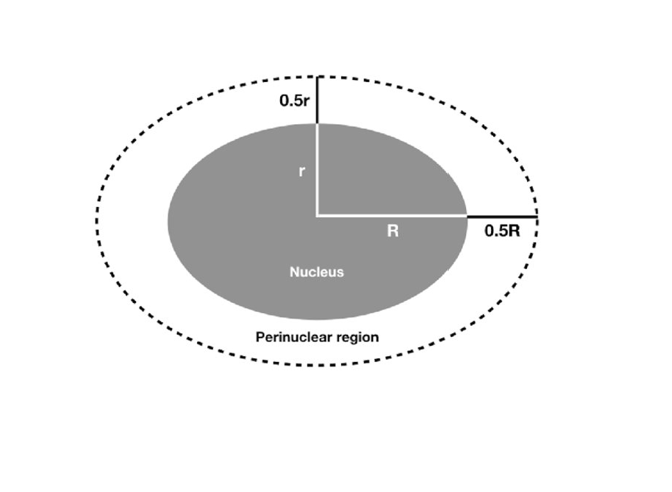
•perinuclear region of cytoplasm
|
•sleep
•regulation of neurotransmitter secretion •positive regulation of transmission of nerve impulse |
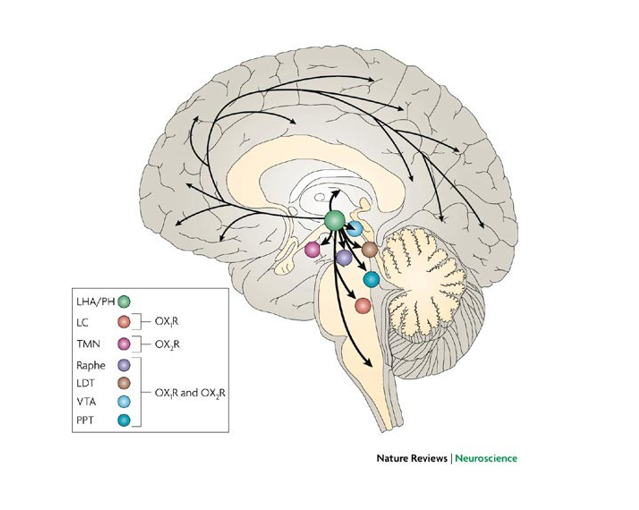
HCRT is located in the perinuclear region of the cytoplasm and is used for neuropeptide hormone activity in the regulation of neurotransmitter secretion and positive regulation of transmission of nerve impulses. Through these mechanisms it is believed that HCRT is involved in circadian rhythm sleep disorders such as Narcolepsy Type 1.
Gaps in Knowledge
The downstream mechanisms of HCRT that regulate circadian rhythm are unclear. HCRT interacts with other types of neurological networks to create a distinct pattern of neuronal activation which we call circadian rhythm however, we are unsure how it does this.
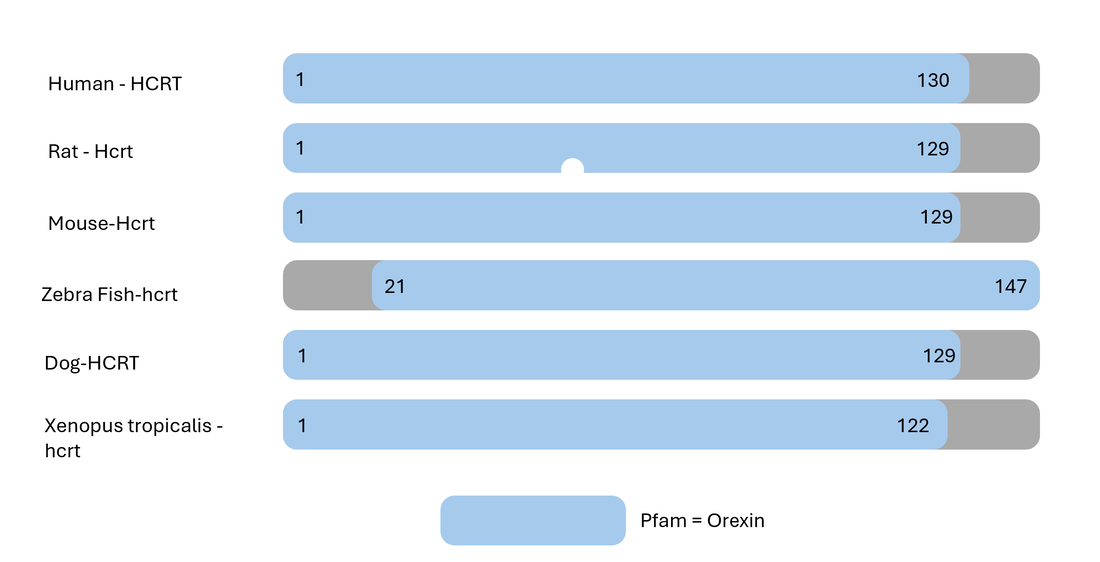
HCRT is conserved among vertebrates
Hypocretin (HCRT) is a precursor protein for Orexin A and B
Danio rerio are great models to study circadian rhythm
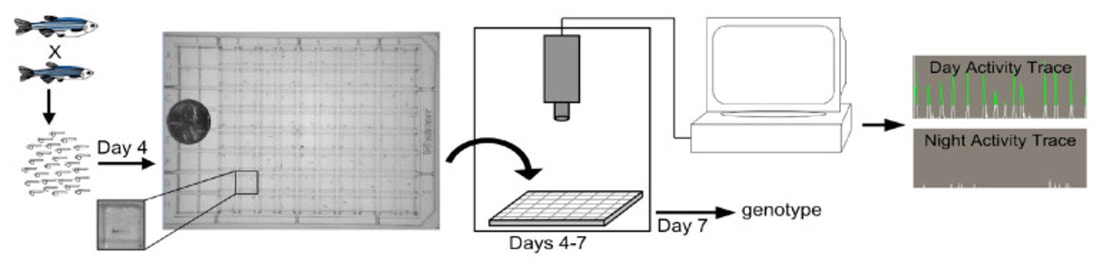
Danio rerio make great model organisms to study how HCRT regulates circadian rhythm for a variety of reasons. First of all, it has a circadian rhythm and homologous genes. They also have small brains that are morphologically similar to humans and less neurons, around 40 for analysis. There is also standardized behavioral analysis software available to track their sleep/wake activity.
HCRT Protein Interactions
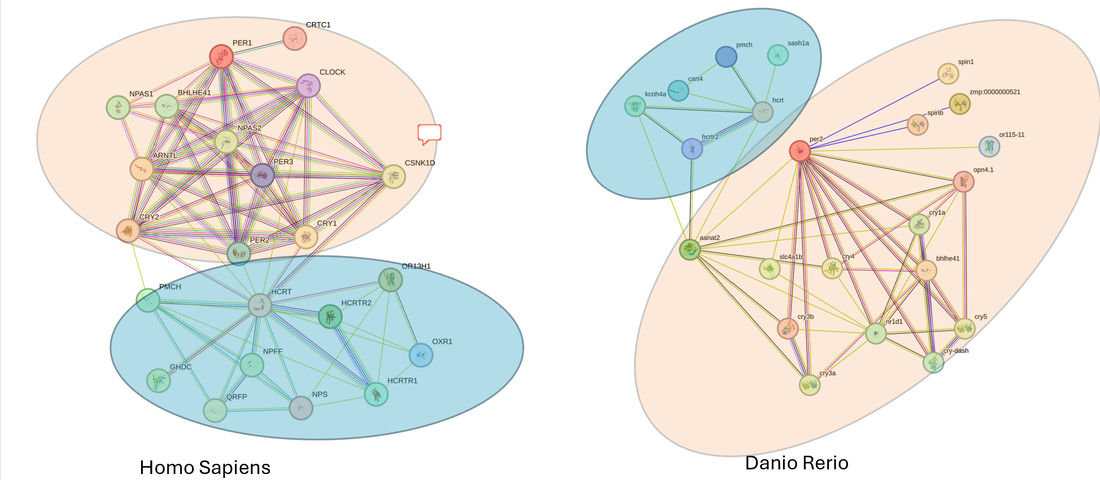
HCRT interacts with proteins involved in Circadian Rhythm and Neuropeptide Signaling. The protein interaction networks between human and Danio rerio HCRT are quite similar. As you can see in the figure below, they are involved in similar GO terms. While the humans have a few more protein interactions, their networks includes similar genes highlighted. Protein interaction networks were obtained from the String database.
Primary Goal
Our primary goal is to elucidate how peptides encoded by HCRT and their binding to HCRTR1 and HCRTR2 contribute to neuronal activation and the proper functioning of the brain's circadian rhythm. I will complete this goal through the following aims:
Aim 1
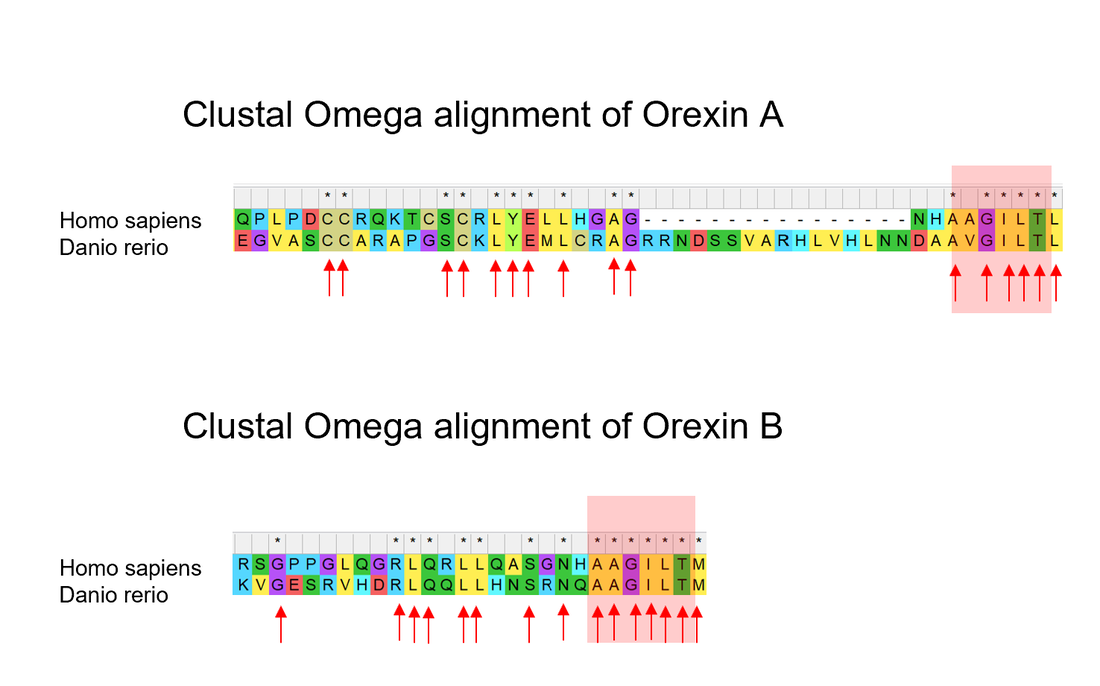
First I will manually align the Orexin A and Orexin B domains in HCRT gene and identify conserved amino acids.
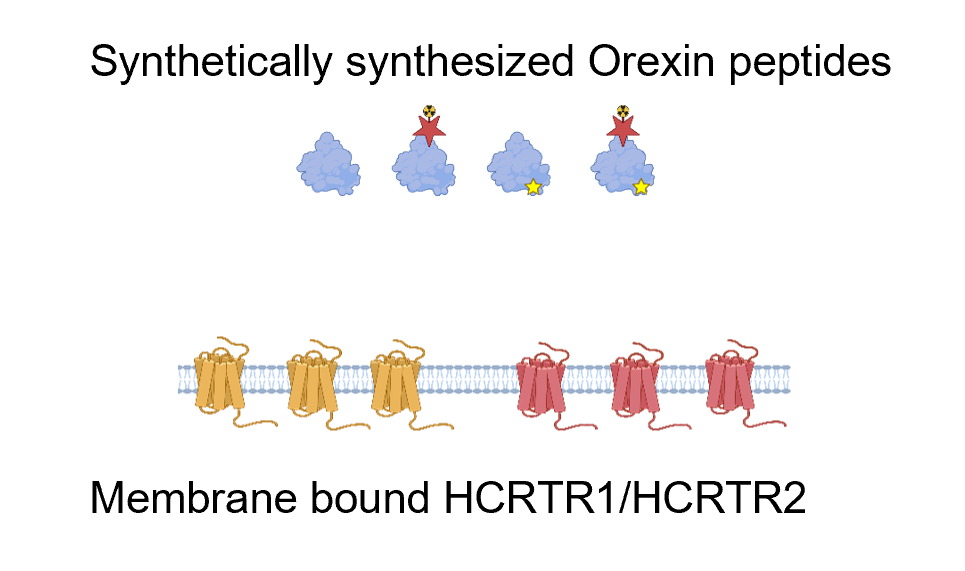
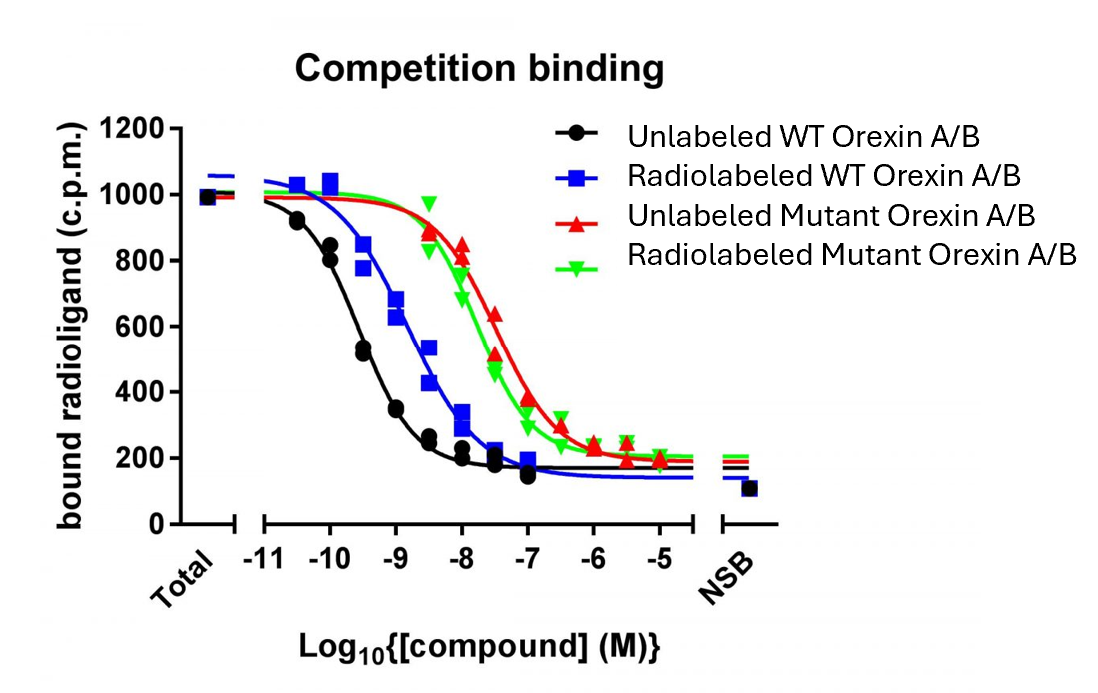
Then I will generate synthetic peptides mutated in the conserved AAs. I will then identifying mutants with a low binding affinity using a radiolabeled competition binding assay to HCRTR1 and/or HCRTR2.
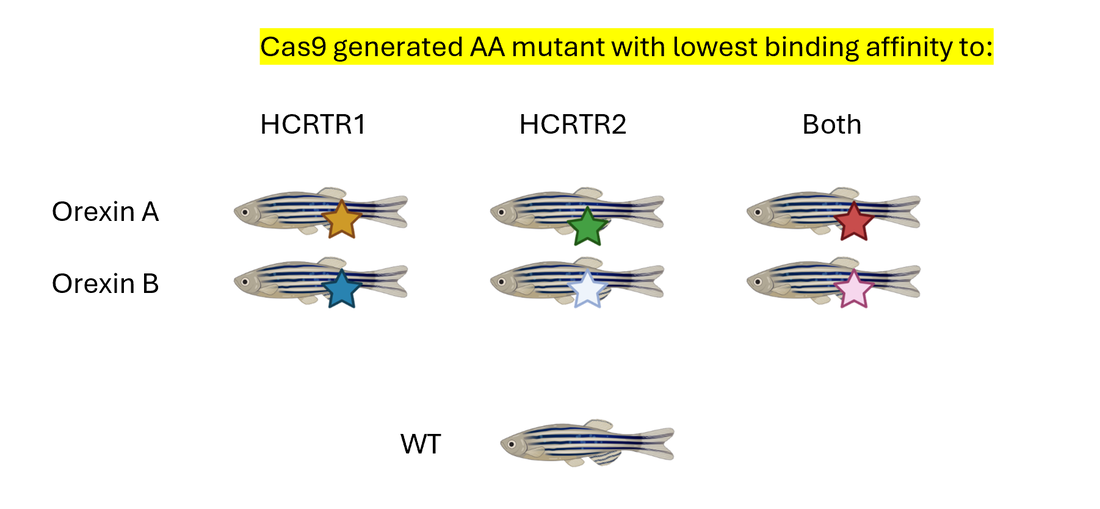
Then I will use CRISPR/Cas9 to generate a mutant library at loci where there was low binding affinity to HCRTR1 and/or HCRTR2 and assess the circadian rhythm phenotype of the of these mutants. I hypothesize that mutants with the lowest binding affinity to HCRTR1 and/or HCRTR2 will have the biggest differences in sleep/wake cycle duration and frequency. Identifying amino acids that impact binding to HCRTR1 and HCRTR2 can give insight into how mutations of HCRT may impact neuronal activation.
Aim 2
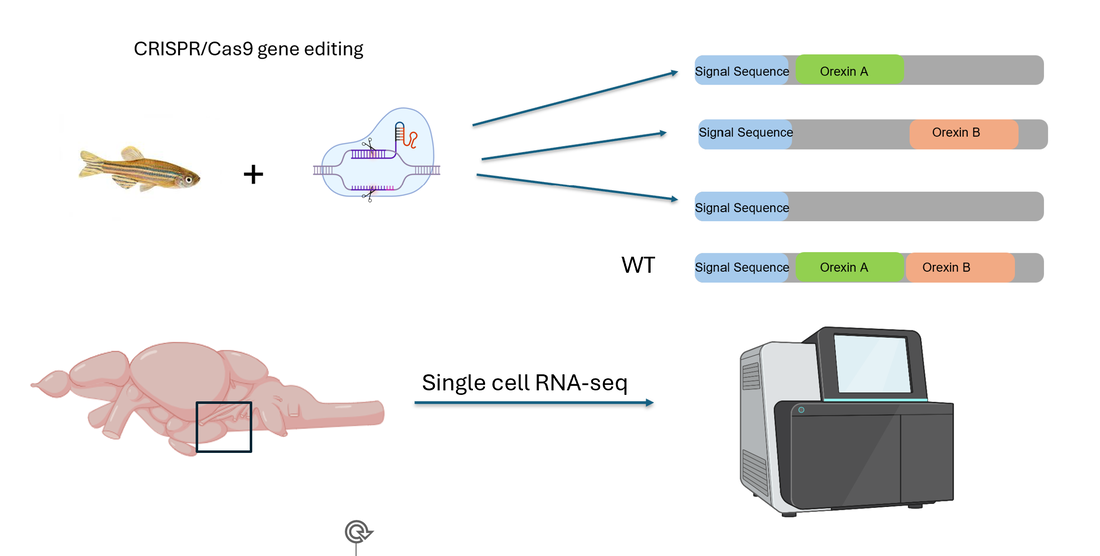
In Aim 2 will perform scRNA seq on Orexin A knockout, Orexin B knockout, and wt mice on mature fish’s dorsolateral hypothalamus tissue after light and dark treatment for each homozygous mutant line of HCRT+wt.
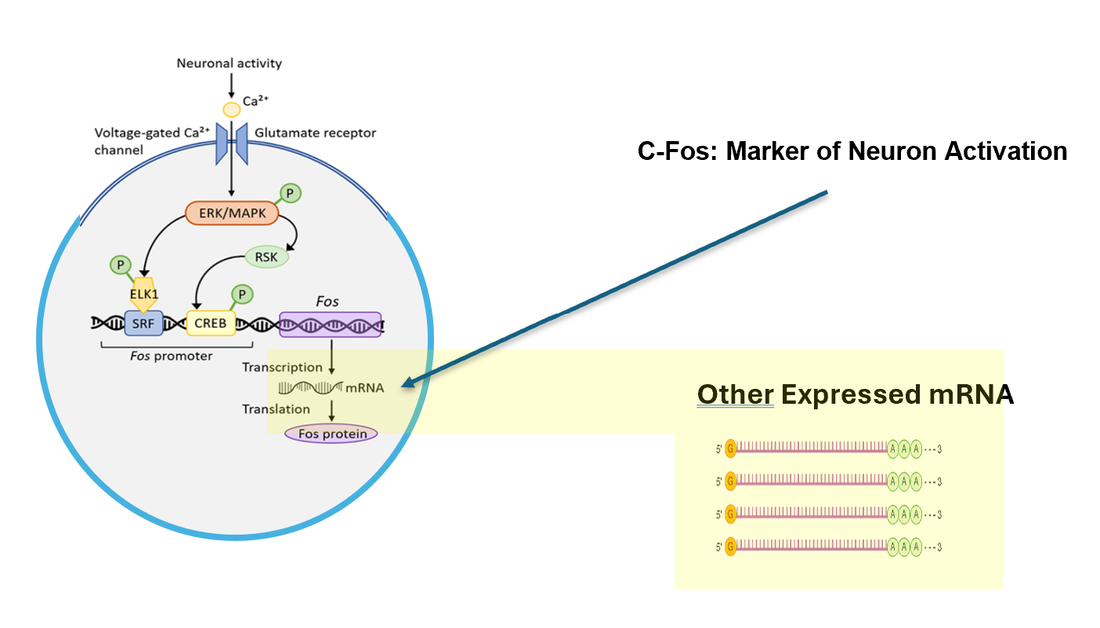
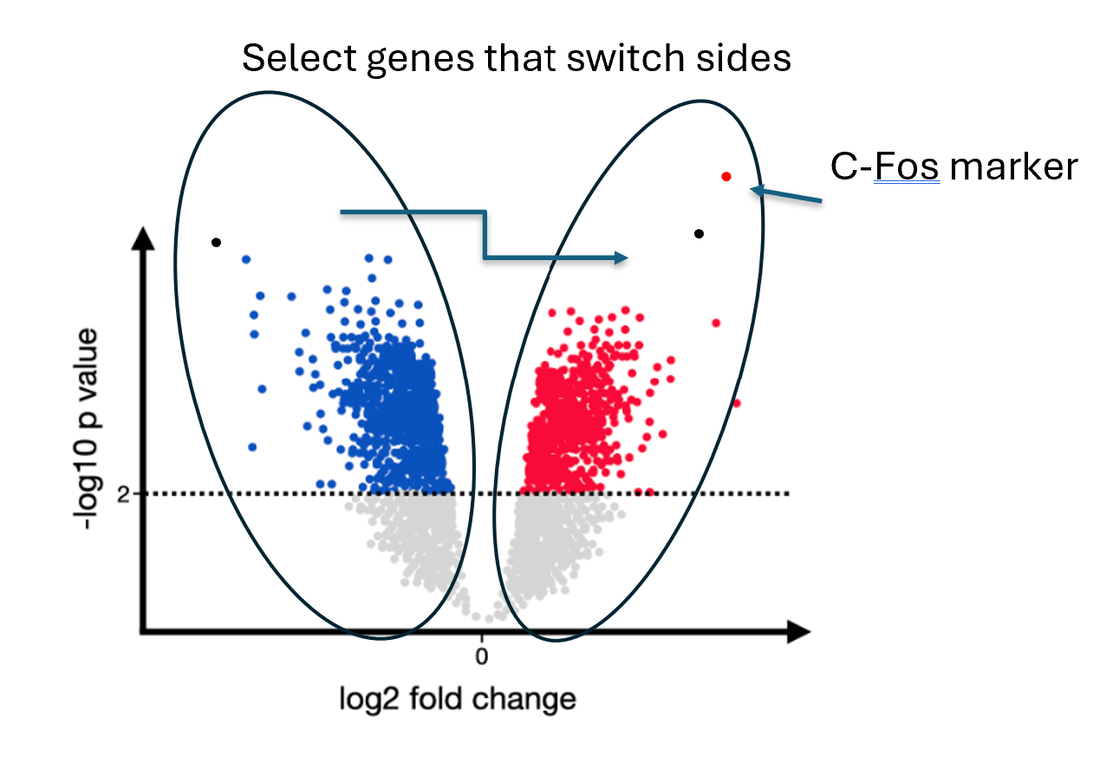
Then I will Identify differentially expressed genes in cell types with high C-Fos expression 1 hour before and after the lights are turned on/off for each mutant. To validate whether a differentially expressed gene is involved in circadian rhythm, I will then knock out and analyze the phenotype of fish where gene expression changed in cells where a C-Fos marker was highly expressed. I hypothesize that knocking out identified differentially expressed genes in activated neurons will highly impact circadian rhythm.
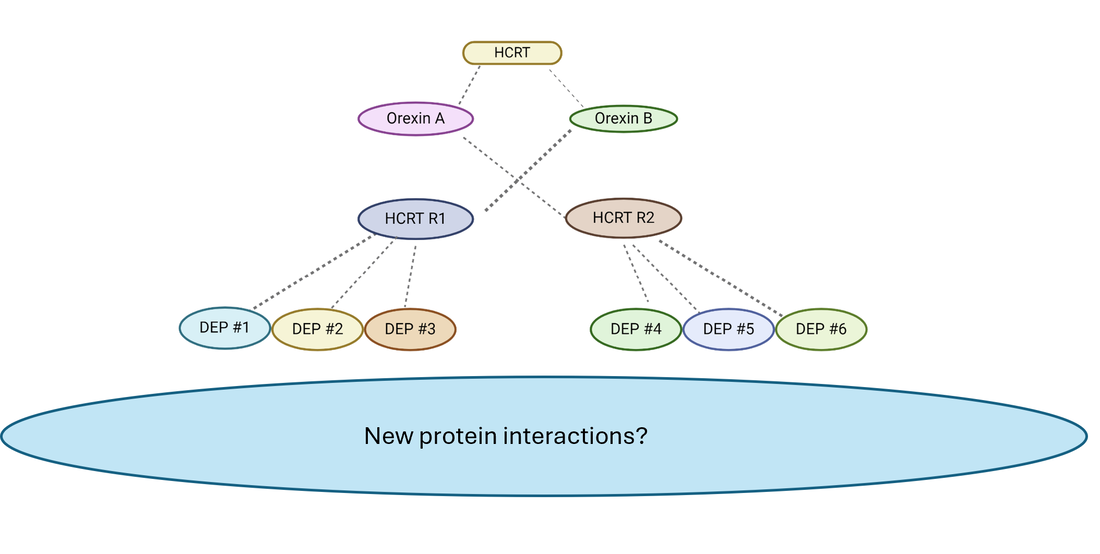
Aim 3
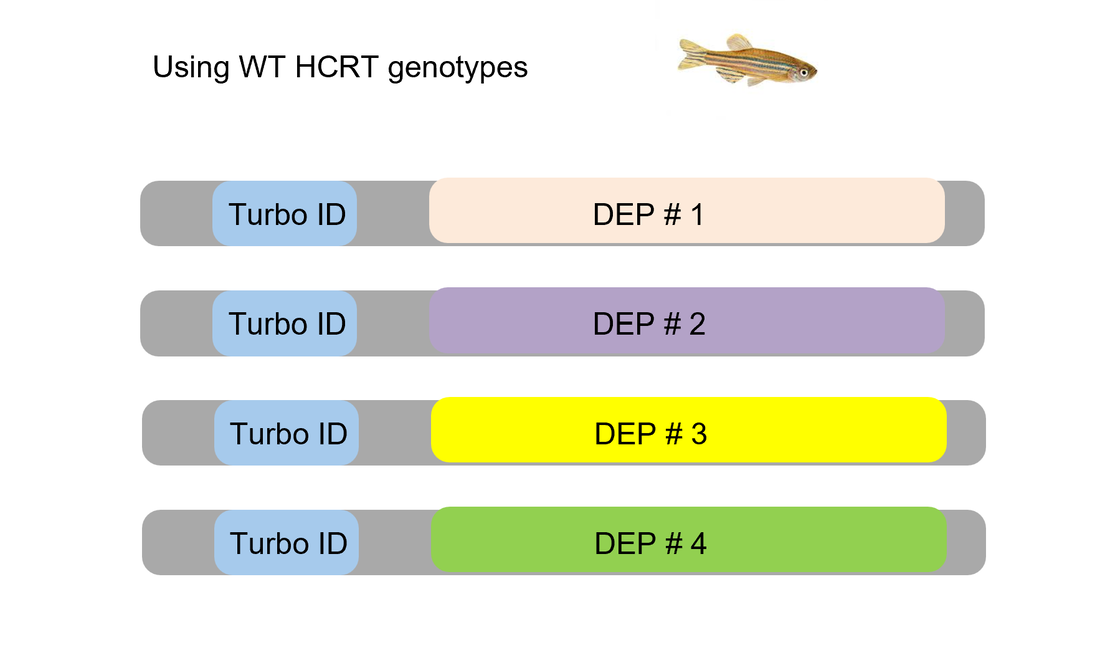
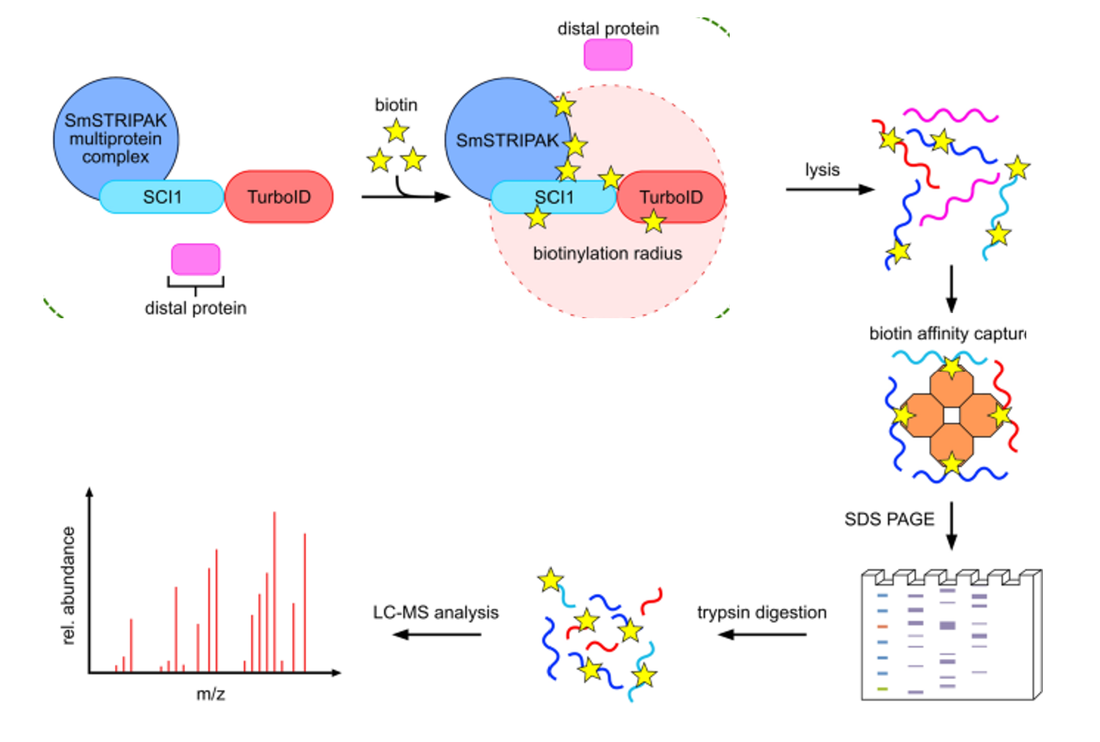
In Aim 3, a TurboID tag will be fused to proteins that were differentially expressed in Aim 2 and showed alterations in circadian rhythm phenotype. Then one hour before and after the lights go off, the brain tissue of the zebrafish will be processed in order to capture what proteins potentially interact with these differentially expressed proteins (DEPs).
After proteins are reconstructed with MaxQuant software, STRING and Pathway Enrichment analysis of proteins in close proximity of these proteins will be performed and connected to other known neurotransmission and regulation-related genes. I hypothesize that STRING analysis will reveal key proteins that are responsible for neuronal activation of the neurons involved in the circadian rhythm.
Conclusions
In conclusion, Narcolepsy Type 1 is both a genetic and autoimmune disorder caused by the lack of HCRT expression in the brain. There is no cure for Narcolepsy Type 1 and HCRTs neuropeptide signaling and circadian rhythm regulation is poorly understood by scientists. By understanding the protein interaction networks in activated neurons, we hope to further understand how HCRT regulates the distinct patterns of neuron activity that we call “circadian rhythm."
Future Directions
In the future it would be useful to begin Identify genetic components that lead to HCRT expression in specific neurons. We should also create a library of HCRT related proteins such as the ones identified in these experiments and screen T cells for autoimmune reactions. In order to asses a possible permanent cure for Narcolepsy Type 1, we should repeat this experiment with an AAV HCRT rescue mouse to see if neuron activation is normal in addition to circadian rhythm.
| giehtbrock2_29_2024.ppt | |
| File Size: | 1666 kb |
| File Type: | ppt |
| giehtbrock3_21_2024_draft2.ppt | |
| File Size: | 1605 kb |
| File Type: | ppt |
| giehtbrock5_8_2024_final_draft.ppt | |
| File Size: | 8615 kb |
| File Type: | ppt |
| giehtbrock5_8_2024_final_draft.pdf | |
| File Size: | 3295 kb |
| File Type: | |
This page was produced as an assignment for Genetics 564, a capstone course at UW-Madison.